Imaging of Matter
New experiment reveals how ultrafast conversion of urea progresses
28 June 2023
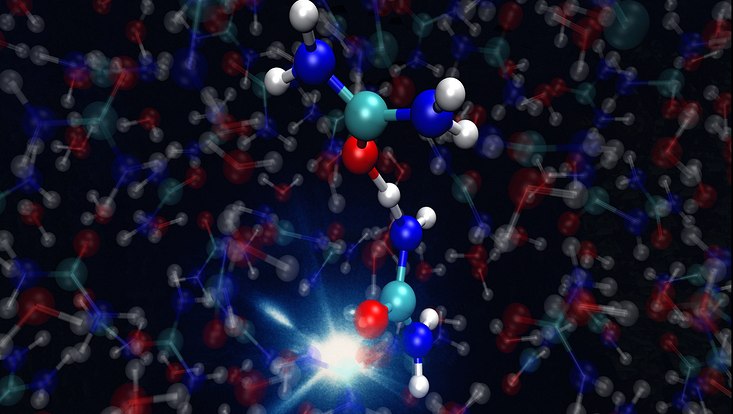
Photo: DESY, Ludger Inhester
Using a special X-ray source, a research team has succeeded in unraveling the first, extremely fast steps of the light-induced urea conversion. With their experiment, the experts were not only able to detect the rapid "proton transaction" between two urea molecules for the first time, but also to determine its duration, as they report in the journal Nature.
How did life begin on Earth? Experts have long been fascinated by this question and over the years have come up with a variety of theories. One hypothesis is that the origin of life can be traced back to warm little ponds which are thought to have existed on Earth four billion years ago. The water in these ponds probably contained urea molecules; these were exposed to ultraviolet radiation from the sun, which at that time would have penetrated to the surface of the earth largely unimpeded. This high-energy radiation was able to convert the urea into reaction products, which then formed biomolecules that later served as the building blocks of life – or so the idea goes.
Theory of "warm little ponds" led to novel experiment
This theory of “warm little ponds” was one of the things that led a research team from Hamburg, Zurich and Geneva to devise and carry out a novel experiment. The team led by Hans Jakob Wörner from the Swiss federal institute of technology ETH Zürich and Jean-Pierre Wolf from the University of Geneva used a special X-ray source to examine the first, extremely rapid steps of light-induced urea conversion.
For their experiment at the University of Geneva, the team shot pulses of a laser beam through a jet of a highly concentrated urea solution. The laser pulses ionized some of the urea molecules, knocking one electron out of each of them. Immediately afterward, the scientists sent an ultra-short pulse of soft X-rays behind them. These acted as a probe, revealing in great detail how the urea reacts to the electron being knocked out. The group then repeated the experiment several times, systematically changing the time interval between the ionizing laser and the X-ray pulses.
As a result, it was possible to reconstruct the sequence of events precisely – down to a few femtoseconds (quadrillionths of a second). Interpreting the resulting spectra proved to be particularly challenging. “This required detailed computer simulations which we developed here at DESY over many years,” explains DESY physicist Ludger Inhester, who works at the Center for Free-Electron Laser Science, a joint venture of DESY, Universität Hamburg and the Max Planck Society.
Observation of fast processes in a molecule in an aqueous environment
The scientists used this setup to work out the initial steps in the conversion of urea. When a urea molecule is ionized, it loses an electron and becomes positively charged. It would like nothing better than to get rid of this positive charge, and that becomes possible when another, non-ionized urea molecule is in the immediate vicinity. “The first molecule then pushes a proton, which is a hydrogen nucleus, over to the neutral molecule,” says Inhester, who also works in the framework of the Cluster of Excellence "CUI: Advanced Imaging of Matter" at Universität Hamburg. “This proton transfer creates a urea radical along with a positively charged urea ion.” Both are chemically reactive and could have led to the formation of RNA molecules billions of years ago – essential building blocks of early life.
“What is new about our experiment is that we were able to observe extremely fast processes in a molecule that exists in an aqueous environment,” explains Inhester points out. “Previous experiments have only looked at reactions like this in the gas phase.” The behavior of molecules suspended in a liquid such as water is particularly relevant to a whole number of questions – especially when it comes to biological processes. Experiments in this type of environment are a challenge not only when it comes to making the measurements, but also in terms of the computations required to interpret the measured data.
In the future, the new technique might also reveal the details of what happens when ionizing radiation strikes tissue, causing radiation damage – the type of research that the new Centre for Molecular Water Science (CMWS) envisages doing, that is currently being set up in international cooperation on the DESY campus. The researchers are also toying with the idea of conducting similar experiments at a far larger X-ray source, the European XFEL in Hamburg. This X-ray laser, which is more than three kilometers long and in which DESY plays a pivotal role, produces the most powerful X-ray pulses in the world. “That would allow us to study this proton transfer from different angles,” says Inhester – who hopes this will reveal additional details about this very fundamental process. Text: DESY, ed.
Reference
Zhong Yin, Yi–Ping Chang, Tadas Balčiūnas, Yashoj Shakya, Aleksa Djorovic, Geoffrey Gaulier, Giuseppe Fazio, Robin Santra, Ludger Inhester, Jean–Pierre Wolf and Hans Jakob Wörner
"Femtosecond Proton Transfer in Urea Solutions Probed by X-ray Spectroscopy"
Nature (2023)